動物低氧工作站 WS-Ox-M
產品描述
在生命科學、基礎醫學及高原生理等研究領域,精確類比低氧環境對於探究缺氧對動物機體的影響至關重要。 低氧工作站正是為此類嚴謹動物實驗而設計的核心設備。
不同於普通低氧實驗箱在操作時必須開箱破壞環境,低氧工作站通過其全密閉結構和內置操作手套,實現了革命性的突破。 研究者無需打開主腔室,即可通過氣密手套在持續、穩定維持目標低氧水準(如1%-21% O₂可調)的環境下,對箱內動物進行各項操作,包括:
Ø日常照料:安全地進行餵食、飲水更換,確保動物長期實驗中的基本生存需求。
Ø實驗干預:執行精準的給葯、注射、手術或生理參數監測(如體溫、心率)。
Ø行為觀察:在恆定低氧條件下進行動物行為學實驗,減少環境波動干擾。
Ø原位采樣:直接在低氧環境中採集血液、組織等樣本,最大程度避免樣本暴露於常氧環境導致的氧化應激或代謝狀態改變,保障後續分析數據的真實性。
Ø設備操作:放置或操作小型實驗儀器(如微型跑步機、攝像頭)。
核心優勢
1、環境穩定性卓越:操作過程「零中斷」低氧環境,氧氣濃度波動極小(通常<±0.1%),為慢性、長期低氧暴露實驗(數天至數月)提供持續、可靠的低氧條件,消除因頻繁開箱導致的濃度回升與恢復延遲問題。
2、操作便利性與連續性:無需等待環境恢復,可隨時、頻繁地進行實驗操作,極大提升實驗效率和靈活性。
3、減少動物應激:密閉操作減少了光線、噪音和人員活動對動物的直接干擾,有助於獲得更接近自然狀態的生理反應數據。
4、保障樣本真實性:低氧環境下的原位採樣和處理,是研究缺氧相關生物標誌物、基因表達和代謝產物的關鍵保障,避免取樣后氧化造成的假像。
5、集成環境控制(可選):高級型號可集成精確的溫濕度控制系統及CO₂清除裝置,為動物提供更舒適、生理狀態更穩定的實驗環境。
6、潔凈度與安全性:密閉設計結合高效過濾系統(HEPA/ULPA),可有效控制微生物污染; 同時為操作者提供物理屏障,隔絕潛在過敏原或實驗性有害物質。
應用場景
低氧工作站是進行高原適應機制研究、缺血性疾病模型、低氧相關腫瘤研究、低氧與代謝疾病的研究、低氧與心血管疾病的研究、圍生期缺血缺氧性腦損傷以及任何要求在穩定低氧環境下進行活體操作或原位取樣的動物實驗的理想平臺。 它克服了傳統低氧箱的核心局限,為獲得嚴謹、可重複的高品質科研數據奠定了堅實的基礎。技術參數
1. 為動物低氧實驗模型的建立提供穩定的低氧環境
2. 按照設定氣體濃度自動配比氣體,維持恆定的氧氣濃度環境。 無需在箱體外混合比例氣體,實驗氧濃度的準確,節省氣源
3. 觸摸屏控制,人性化介面,操作簡單
4. 監測參數:溫度、濕度、氧氣濃度、二氧化碳濃度
5. 控制精度:±0.1%
6. 非色散紅外(NDIR)二氧化碳感測器,測量範圍:0~5000ppm
7. 進口電化學氧氣濃度檢測器,測量範圍:0-25%vol(可選配0.1-99.0%),線性度好,檢測準確、使用壽命長。 具有溫度補償機制
8. 溫度檢測:進口高精度溫度感測器
9. 氧氣濃度變化動態曲線,直觀瞭解氧氣濃度變化的過程
10. 內置紫外滅菌燈,可定時滅菌
11. 具有定時功能,實驗完成,自動恢復常氧狀態,並伴有聲音提示
12. 氧氣濃度自動校準:通過控制器對感測器快速校準
13. 特有的氣體混合及循環機制,保證箱體內氣體濃度的均一
14. 高性能電磁閥,性能穩定,超長壽命
15. 前面板可徒手拆卸,便於放置設備
可選配功能
1、溫度濕度控制功能,控溫範圍:室溫+3~45°C(可選4~45°C),調節精度0.1°C;濕度40~85%RH
2、遠端監控:可通過電腦、手機遠端監控實驗運行狀態,具有可夜視紅外攝像頭
3、二氧化碳吸附裝置
4、二氧化碳濃度控制功能,可設定目標濃度,控制範圍:0-20.0%,其他範圍可選
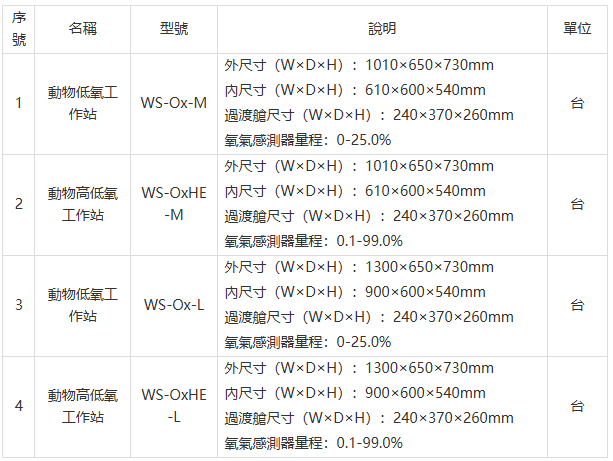
型號選擇
相關文獻
[1] Drekolia M K, Mettner J, Wang D, et al. Cystine import and oxidative catabolism fuel vascular growth and repair via nutrient-responsive histone acetylation[J]. Cell Metabolism (IF 30.9), 2025.
[2] Wu L W, Chen M, Jiang C Y, et al. Inactivation of AXL in Cardiac Fibroblasts Alleviates Right Ventricular Remodeling in Pulmonary Hypertension[J]. Advanced Science (IF 14.1), 2025: e08995.
[3] Lei R, Gu M, Li J, et al. Lipoic acid/trometamol assembled hydrogel as injectable bandage for hypoxic wound healing at high altitude[J]. Chemical Engineering Journal (IF 13.4), 2024, 489: 151499.
[4] Li Z, Li H, Qiao W, et al. Multi-omics dissection of high TWAS-active endothelial pathogenesis in pulmonary arterial hypertension: bridging single-cell heterogeneity, machine learning-driven biomarkers, and developmental reprogramming[J]. International Journal of Surgery (IF 10.1), 10.1097.
[5] Pei Y, Huang L, Wang T, et al. Bone marrow mesenchymal stem cells loaded into hydrogel/nanofiber composite scaffolds ameliorate ischemic brain injury[J]. Materials Today Advances (IF 10), 2023, 17: 100349.
[6] Wang Q, Liu J, Li R, et al. Macrophage κ-opioid receptor inhibits hypoxic pulmonary hypertension progression and right heart dysfunction via an SCD1-dependent anti-inflammatory response[J]. Genes & Diseases (IF 9.4), 2025: 101604.
[7] Wang Y, Zhang R, Chen Q, et al. PPARγ Agonist Pioglitazone Prevents Hypoxia-induced Cardiac Dysfunction by Reprogramming Glucose Metabolism[J]. International Journal of Biological Sciences, 2024, 20(11): 4297.
[8] Wang Y, Shen P, Wu Z, et al. Plasma Proteomic Profiling Reveals ITGA2B as a key regulator of heart health in high-altitude settlers[J]. Genomics, Proteomics & Bioinformatics, 2025: qzaf030.
[9] Lan Y, Zhao S, Song Y, et al. Physicochemical properties of selenized quinoa protein hydrolysate and its regulatory effects on neuroinflammation and gut microbiota in hypoxic mice[J]. Journal of Future Foods, 2025.
[10] Pan Z, Yao Y, Liu X, et al. Nr1d1 inhibition mitigates intermittent hypoxia-induced pulmonary hypertension via Dusp1-mediated Erk1/2 deactivation and mitochondrial fission attenuation[J]. Cell Death Discovery, 2024, 10(1): 459.
[11] Zhou Y, Ni Z, Liu J, et al. Gut Microbiota‐Associated Metabolites Affected the Susceptibility to Heart Health Abnormality in Young Migrants at High‐Altitude: Gut Microbiota and Associated Metabolites Impart Heart Health in Plateau[C]//Exploration. 2025: 20240332.
[12] Li C, Zhao Z, Jin J, et al. NLRP3-GSDMD-dependent IL-1β Secretion from Microglia Mediates Learning and Memory Impairment in a Chronic Intermittent Hypoxia-induced Mouse Model[J]. Neuroscience, 2024, 539: 51-65.
[13] Yang W, Li M, Ding J, et al. High-altitude hypoxia exposure inhibits erythrophagocytosis by inducing macrophage ferroptosis in the spleen[J]. Elife, 2024, 12: RP87496.
[14] You Z, Huang Q, Zeng L, et al. Rab26 promotes hypoxia-induced hyperproliferation of PASMCs by modulating the AT1R-STAT3-YAP axis[J]. Cellular and Molecular Life Sciences, 2025, 82(1): 1-16.
[15] Pei C, Shen Z, Wu Y, et al. Eleutheroside B Pretreatment Attenuates Hypobaric Hypoxia‐Induced High‐Altitude Pulmonary Edema by Regulating Autophagic Flux via the AMPK/mTOR Pathway[J]. Phytotherapy Research, 2024, 38(12): 5657-5671.
[16] Duan H, Han Y, Zhang H, et al. Eleutheroside B Ameliorates Cardiomyocytes Necroptosis in High-Altitude-Induced Myocardial Injury via Nrf2/HO-1 Signaling Pathway[J]. Antioxidants, 2025, 14(2): 190.
[17] Song J, Zheng J, Li Z, et al. Sulfur dioxide inhibits mast cell degranulation by sulphenylation of galectin-9 at cysteine 74[J]. Frontiers in Immunology, 2024, 15: 1369326.
[18] Jia N, Shen Z, Zhao S, et al. Eleutheroside E from pre-treatment of Acanthopanax senticosus (Rupr. etMaxim.) Harms ameliorates high-altitude-induced heart injury by regulating NLRP3 inflammasome-mediated pyroptosis via NLRP3/caspase-1 pathway[J]. International Immunopharmacology, 2023, 121: 110423.
[19] Huang Q, Han X, Li J, et al. Intranasal Administration of Acetaminophen-Loaded Poly (lactic-co-glycolic acid) Nanoparticles Increases Pain Threshold in Mice Rapidly Entering High Altitudes[J]. Pharmaceutics, 2025, 17(3): 341.
[20] Wu Y, Tang Z, Du S, et al. Oral quercetin nanoparticles in hydrogel microspheres alleviate high-altitude sleep disturbance based on the gut-brain axis[J]. International Journal of Pharmaceutics, 2024, 658: 124225.
[21] Zhou Z, Zhao Q, Huang Y, et al. Berberine ameliorates chronic intermittent hypoxia‐induced cardiac remodelling by preserving mitochondrial function, role of SIRT6 signalling[J]. Journal of Cellular and Molecular Medicine, 2024, 28(12): e18407.
[22] Shang W, Huang Y, Xu Z, et al. The impact of a high-carbohydrate diet on the cognitive behavior of mice in a low-pressure, low-oxygen environment[J]. Food & Function, 2025, 16(3): 1116-1129.
[23] Pei C, Jia N, Wang Y, et al. Notoginsenoside R1 protects against hypobaric hypoxia-induced high-altitude pulmonary edema by inhibiting apoptosis via ERK1/2-P90rsk-BAD ignaling pathway[J]. European Journal of Pharmacology, 2023, 959: 176065.
[24] Xie L, Wu Q, Huang H, et al. Neuroregulation of histamine of circadian rhythm disorder induced by chronic intermittent hypoxia[J]. European Journal of Pharmacology, 2025: 177662.
[25] Ding Y, Liu W, Zhang X, et al. Bicarbonate-Rich Mineral Water Mitigates Hypoxia-Induced Osteoporosis in Mice via Gut Microbiota and Metabolic Pathway Regulation[J]. Nutrients, 2025, 17(6): 998.
[26] Gu N, Shen Y, He Y, et al. Loss of m6A demethylase ALKBH5 alleviates hypoxia-induced pulmonary arterial hypertension via inhibiting Cyp1a1 mRNA decay[J]. Journal of Molecular and Cellular Cardiology, 2024.
[27] Luan X, Zhu D, Hao Y, et al. Qibai Pingfei Capsule ameliorated inflammation in chronic obstructive pulmonary disease (COPD) via HIF-1 α/glycolysis pathway mediated of BMAL1[J]. International Immunopharmacology, 2025, 144: 113636.
[28] Jiang H, Lu C, Wu H, et al. Decreased cold‐inducible RNA‐binding protein (CIRP) binding to GluRl on neuronal membranes mediates memory impairment resulting from prolonged hypobaric hypoxia exposure[J]. CNS Neuroscience & Therapeutics, 2024, 30(9): e70059.
[29] Chang P, Xu M, Zhu J, et al. Pharmacological Inhibition of Mitochondrial Division Attenuates Simulated High‐Altitude Exposure‐Induced Memory Impairment in Mice: [30] Involvement of Inhibition of Microglia‐Mediated Synapse Elimination[J]. CNS Neuroscience & Therapeutics, 2025, 31(6): e70473.
[30] Liu C, Qu D, Li C, et al. miR‐448‐3p/miR‐1264‐3p Participates in Intermittent Hypoxic Response in Hippocampus by Regulating Fam76b/hnRNPA2B1[J]. CNS Neuroscience & Therapeutics, 2025, 31(2): e70239.
[31] Wu L W, Chen M, Jiang D J, et al. TCF7 enhances pulmonary hypertension by boosting stressed natural killer cells and their interaction with pulmonary arterial smooth muscle cells[J]. Respiratory Research, 2025, 26(1): 202.
[32] Xie L, Wu Q, Huang H, et al. Neuroregulation of histamine of circadian rhythm disorder induced by chronic intermittent hypoxia[J]. European Journal of Pharmacology, 2025: 177662.
[33] Cai S, Li Z, Bai J, et al. Optimized oxygen therapy improves sleep deprivation-induced cardiac dysfunction through gut microbiota[J]. Frontiers in Cellular and Infection Microbiology, 2025, 15: 1522431.
[34] Wang X, Xie Y, Niu Y, et al. CX3CL1/CX3CR1 signal mediates M1-type microglia and accelerates high-altitude-induced forgetting[J]. Frontiers in Cellular Neuroscience, 2023, 17: 1189348.
[35] He Y, Wang Y, Duan H, et al. Pharmacological targeting of ferroptosis in hypoxia-induced pulmonary edema: therapeutic potential of ginsenoside Rg3 through activation of the PI3K/AKT pathway[J]. Frontiers in Pharmacology, 2025, 16: 1644436.
[36] Guo Y, Qin J, Sun R, et al. Molecular hydrogen promotes retinal vascular regeneration and attenuates neovascularization and neuroglial dysfunction in oxygen-induced retinopathy mice[J]. Biological Research, 2024, 57.
[37] Liu L, Zhang J, Song S, et al. Paraventricular nucleus neurons: important regulators of respiratory movement in mice with chronic intermittent hypoxia[J]. Annals of Medicine, 2025, 57(1): 2588664.
[38] Ma Q, Ma J, Cui J, et al. Oxygen enrichment protects against intestinal damage and gut microbiota disturbance in rats exposed to acute high-altitude hypoxia[J]. Frontiers in Microbiology, 2023, 14.
[39] Lan J, Lin J, Guo Y, et al. Sequencing and bioinformatics analysis of exosome-derived miRNAs in mouse models of pancreatic injury induced by OSA[J]. Frontiers in Physiology, 2025, 16: 1712442.
[40] Feng X, Li C, Zhang W, et al. Mechanism of retinal angiogenesis induced by HIF-1α and HIF-2α under hyperoxic conditions[J]. Scientific Reports, 2025, 15(1): 36049.
[41] Yao Y, Chen Y, Li Y, et al. TGM2 Enhances Hypobaric Hypoxia-mediated Brain Injury Via Regulating NLRP3/GSDMD Signaling[J]. Neurochemical Research, 2025, 50(6): 1-11.
[42] Yang A, Guo L, Zhang Y, et al. MFN2-mediated mitochondrial fusion facilitates acute hypobaric hypoxia-induced cardiac dysfunction by increasing glucose catabolism and ROS production[J]. Biochimica et Biophysica Acta (BBA)-General Subjects, 2023: 130413.
[43] Chu H, Jiang W, Zuo N, et al. Astrocyte activation: A key mediator underlying chronic intermittent hypoxia-induced cognitive dysfunction[J]. Sleep Medicine, 2025: 106692.
[44] Xu A, Huang F, Chen E, et al. Hyperbaric oxygen therapy attenuates heatstroke-induced hippocampal injury by inhibiting microglial pyroptosis[J]. International Journal of Hyperthermia, 2024, 41(1): 2382162.
[45] Zhang Z, Zheng X, He Y, et al. Hyperbaric oxygen ameliorates neuroinflammation in heat-stressed BV-2 microglial cells: potential involvement of EAAT2 regulation[J]. International Journal of Hyperthermia, 2025, 42(1): 2583133.
[46] Jinyu F, Huaicun L, Yanfei Z, et al. Nogo-A Protein Mediates Oxidative Stress and Synaptic Damage Induced by High-altitude Hypoxia in the Rat Hippocampus[J

